NUR-SULTAN – The Kazakh Biosafety Research Institute presented the QazVac vaccine that researchers have been developing over the last year, reports KazInform news agency.
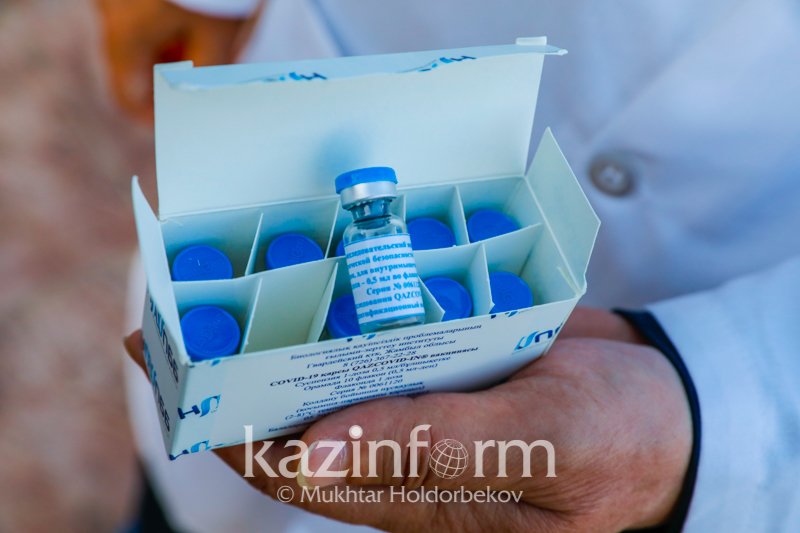
Vaccine vials. Photo credit: Kazinform/Mukhtar Holdorbekov
“This vaccine was developed by our institute. There are 10 vials in the box, which make 10 doses. Each vial contains one dose. The vial contains half a milliliter of vaccine or 10 drops. It needs to be shaken well before use,” said one of vaccine developers Lespek Kutumbetov.
The same vial is used to inject the second component 21 days later.
“The vaccine is stored in a normal domestic refrigerator at two to eight degrees Celsius. It cannot be frozen and it can be stored for a year,” said Kutumbetov.
The first 50,000 doses of the vaccine produced in Kazakhstan will be available by the end of April.

Lespek Kutumbetov. Photo credit: Kazinform/Mukhtar Holdorbekov
“In March 2020, the first coronavirus case was confirmed in Kazakhstan. On March 23, samples were taken from those who became ill, and biological properties, from which a virus identified as COVID-19 was isolated, were studied,” said the Kazakh Ministry of Education and Science in a statement.
The vaccine was registered by the World Health Organization as a candidate vaccine on May 15, 2020. The phase I clinical trials in September 2020 involved 44 volunteers, while a phase II clinical trial began on Oct. 17 with 200 volunteers.
On Dec. 19, the Kazakh Ministry of Healthcare granted permission for phase III clinical trials. 3000 people volunteered and were vaccinated with the QazCovid-in vaccine in the cities of Almaty and Taraz. No severe side effects or complaints were reported.
“The exact date of completion of the third phase of clinical trials is July 9, 2021. After that, there will be a lot of work to process the data from 3,000 volunteers, which will take around 20 calendar days. After that our report will be submitted to the Ministry of Healthcare for thorough consideration in accordance with certain rules and regulations,” said head of the research institute Kunsulu Zakarya.
